Bioavailability of Liposomal Vitamin C in Powder Form
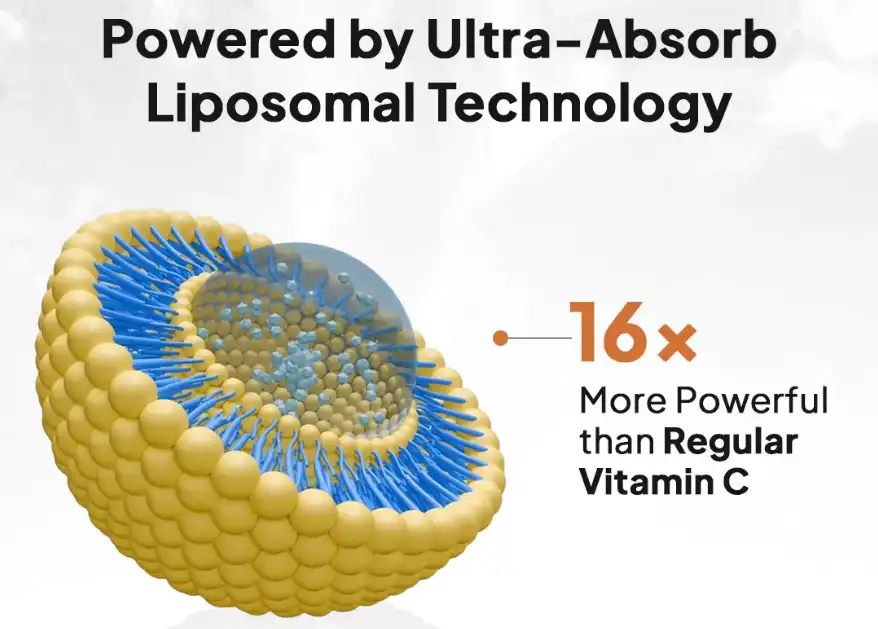
Bioavailability of liposomal vitamin C in powder form is a big step forward in the science of nutritional supplements; it allows cells to absorb it better than regular ascorbic acid. Putting vitamin C inside phospholipid bilayers makes microscopic delivery systems that get around digestive hurdles. This means that up to 90% of the supplement is absorbed, compared to only 20% with regular supplements. Because powder formulations are more bioavailable, they are very useful for business-to-business uses that need consistent therapeutic results and precise dosage.
Understanding Liposomal Vitamin C and Its Bioavailability
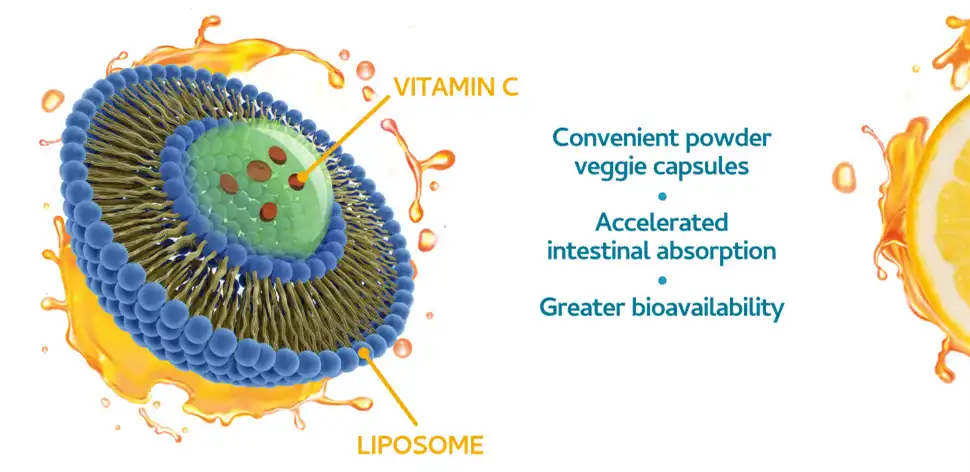
The encapsulation technology that converts water-soluble vitamin C into a lipid-compatible delivery method is the primary distinction between liposomal vitamin C and regular ascorbic acid. This new idea fixes the main problem with regular vitamin C supplements: they don't work well with cells because water-soluble nutrients don't mix well with lipid-based cell walls.
The Science Behind Enhanced Absorption
Bioavailability is a way to measure how well nutrients get to the cells they need to have biological effects. Traditional vitamin C has a hard time being absorbed in the digestive track; only 20–30% of the doses that are eaten get into the bloodstream. The phospholipid encapsulation in liposomal products makes protective vesicles that blend in with the cell membranes of the intestines, making it easier for nutrients to get to the cells directly.
Researchers have found that liposomal packaging raises the amount of vitamin C in the blood by 135% to 200% compared to the same amounts of ascorbic acid. Liposomes make this improvement possible because they keep stomach enzymes from breaking down vitamin C and let it pass through cell walls without being broken down.
Powder Form Advantages in Bioavailability
Liposomal vitamin C in powder form has special advantages over liquid forms in terms of absorption. The dehydrated liposomal structure stays solid until it is reconstituted. This keeps it from breaking down too quickly, which often happens with liquid preparations. When the powder comes into touch with gastric fluids, it naturally forms intact liposomal suspensions. This makes sure that the nutrients are delivered optimally at the time of consumption.
The powder has a high surface area, allowing liposomes to rapidly rehydrate and form functional vesicles. This helps achieve more consistent bioavailability across different doses. Consistent absorption kinetics ensure reliable product performance and regulatory compliance in B2B applications.
Comparing Liposomal Vitamin C Powder with Other Vitamin C Forms
The market for liposomal vitamin C powder supplements includes a wide range of different formulations, each with its own absorption properties and business factors. Knowing these differences helps you make smart purchasing choices that are in line with your product development goals and the needs of your target market.
Absorption Efficiency Analysis
Ascorbic acid tablets and capsules normally give vitamin C through passive diffusion systems that stop working when the concentration is too low. Studies show that as the amount goes above 200 mg, the rate of absorption slows down, and by 1000 mg, the drug is less than 50% bioavailable. Because of this limitation, dosing plans have to be changed often, which may make it harder for people to follow them.
Liposomal vitamin C powder gets around these problems by using active transport systems that keep absorption rates the same at different dose levels. In clinical studies, plasma vitamin C levels stayed high for 6–8 hours after a single dose, while they only stayed high for 2–4 hours with regular supplements.
Formulation Stability Considerations
There are problems with the stability of liquid liposomal products that affect both their bioavailability and their ability to make money. Light, changes in temperature, and oxidative conditions can damage liposomes, which makes therapies less effective over time. The needs for manufacturing and storing liquid products often make them more expensive to make and harder to get to customers.
These worries are taken care of by powder forms, which use anhydrous processing to keep the liposomal structure forever as long as they are stored properly. The powder has a long shelf life and is easy to handle, which makes it perfect for buying in bulk and shipping internationally, which are popular in B2B markets.
Gastrointestinal Tolerance Profile
When taken in therapeutic amounts above 500 mg, traditional vitamin C supplements can irritate the stomach, make you sick, and cause osmotic diarrhea. These side effects happen because ascorbic acid is acidic and the stomach empties quickly, which makes it hard for the intestines to absorb it.
Liposomal encapsulation greatly lowers the negative effects on the digestive system by keeping vitamin C from coming into direct touch with the stomach lining and allowing its controlled, gradual release throughout the digestive system. This better tolerance profile increases the number of people who can buy high-dose vitamin C products, which helps premium positioning tactics.
Advantages of Liposomal Vitamin C Powder for B2B Applications
Beyond improved bioavailability, liposomal vitamin C powder offers operational efficiencies and market positioning possibilities that help you stand out from the competition. These benefits are in line with current industry trends that stress scientific proof and better health results for consumers.
Manufacturing and Logistics Benefits
Powder formulations make production more efficient by making it easier to mix, package, and check the quality of the product. The ingredient's chemical stability and ability to flow well make it useful for automated production systems. It also lowers the risk of contamination that comes with handling liquids. On a commercial level, these operational benefits mean that production is more efficient and costs are lower.
Compared to liquid alternatives, the powder's small size makes shipping amounts more efficient and cuts down on transportation costs. Because powder versions are easier to ship internationally and don't need as much paperwork, international shipping rules favor them. These benefits in terms of logistics are especially useful for managing global supply chains and trying to grow markets.
Versatility in Product Development
Liposomal vitamin C powder's useful qualities open up a wide range of product applications. The ingredient can be added to instant drink mixes by beverage makers to provide liposomal effects right when the drinks are drunk. Nutraceutical companies can make high-potency capsules that last longer and have bioavailability patterns that can be predicted.
The powder can improve the stability of vitamin C in topical products and help them penetrate deeper into the skin, which is useful for cosmetic uses. Because of this, B2B partners can use the same ingredient in more than one product line, which makes purchasing and managing supplies more efficient.
Market Positioning and Consumer Education
The scientific basis for liposomal vitamin C makes marketing stories more interesting to health-conscious people looking for nutritional solutions based on proof. Clinical data showing better absorption and effectiveness backs up premium pricing tactics and claims that a product is different.
Bioavailability ideas are becoming more and more popular among consumers, which is driving the need for more advanced delivery methods. B2B partners can make money off of this trend by marketing liposomal vitamin C products as scientifically better alternatives to regular supplements, backed up by clear data on their effectiveness and educational material.
Selecting and Procuring Quality Liposomal Vitamin C Powder
When buying liposomal vitamin C powder, it's important to carefully look at the skills of the supplier, the product specifications, and the quality assurance systems. Because making liposomes is so complicated, vendors must go through strict qualification processes to make sure that the products always work well and meet legal requirements.
Supplier Evaluation Criteria
Suppliers of liposomal vitamin C should have a thorough knowledge of phospholipid chemistry and cutting-edge encapsulation techniques. Manufacturing facilities should keep their ISO 22000, GMP, and HACCP licenses and use approved analytical methods to test the stability and characterisation of liposomes.
When choosing a supplier, intellectual property is very important because proprietary encapsulation technologies often decide how well a product works and how long it can stay on the market. For difficult product development projects, suppliers who offer full technical help, such as formulation advice and stability studies, are more valuable.
Analytical Specifications and Testing
Some important quality factors for liposomal vitamin C powder are the range of particle sizes, how well the powder encapsulates, the amount of vitamin C it contains, and its bacterial safety. For best bioavailability performance, acceptable standards usually call for 85–95% capsule efficiency and mean particle sizes between 50 and 200 nanometers.
Standardized dissolution testing should be part of stability testing methods, along with studies of accelerated aging, moisture sensitivity, and bioavailability. Suppliers who offer full analytical reports of analysis and stability data show that they are dedicated to quality control and following the rules.
Regulatory and Compliance Considerations
Different countries have different rules about liposomal vitamin C goods, which affects what they can say on the label, how they are made, and the paperwork needed to import them. Suppliers with a history of regulatory knowledge and global registration support make it easier to get into markets while lowering the risk of not complying.
Traceability systems that allow full supply chain paperwork from sourcing raw materials to releasing the finished product make sure that the company is ready for audits and can recall products if needed. For pharmaceutical uses and markets with strict quality standards, these systems are important.
Best Practices for Using and Marketing Liposomal Vitamin C Powder
To successfully bring liposomal vitamin C products to market, formulation optimization, market positioning, and customer education must all be done in a planned way. These best practices make the most of the solubility benefits of the ingredient while also building long-term competitive advantages.
Formulation Optimization Strategies
Liposomal vitamin C powder should be taken in amounts of 500 to 1500 mg per day, spread out over several doses to keep plasma levels steady. To improve absorption rates and therapeutic windows, product makers should think about time-release mechanisms or enteric coating technologies.
Combinations of ingredients that work well together can make vitamin C work better while also giving you chances to market your product in a unique way. Bioflavonoids, especially quercetin and rutin, help the body recycle vitamin C and fight free radicals. Mineral cofactors, like zinc and selenium, work with immune support uses.
Consumer Education and Market Communication
It is important for educational programs to focus on the scientific reasons behind liposomal delivery and stay away from overly technical answers that could confuse people. Visual aids that show how cells absorb substances and compare bioavailability data effectively explain the benefits of a product.
Validation from a third party, like clinical studies and expert endorsements, boosts trustworthiness and helps with premium positioning strategies. When you work together with nutritionists and health care professionals, you build real advocacy channels that affect people's decisions about what to buy.
Success Stories and Market Applications
Leading nutraceutical businesses have had a lot of success in the market by marketing liposomal vitamin C goods as high-end alternatives to regular supplements. One well-known example is a company that makes specialty supplements that saw 300% growth in sales in just 18 months after releasing liposomal vitamin C pills for immune support markets.
Functional beverage uses show a lot of promise. For example, several brands have successfully launched liposomal vitamin C drink mixes that sell for more than regular vitamin C products. These examples of success show how the market could benefit from new liposomal uses in a range of product types.
Conclusion
The bioavailability benefits of liposomal vitamin C powder make it a great choice for business partners looking for healthy ingredients that have been proven to work by science. The better absorption properties, along with the higher steadiness and wide range of possible uses, make this technology the best choice for tough market situations. Manufacturing efficiencies and customer appeal factors help keep a business competitive while meeting the growing need for health solutions based on evidence. B2B partners can take advantage of these benefits and build lasting market differentiation through strategic sourcing and formulation optimization.
FAQ
What makes liposomal vitamin C powder more bioavailable than traditional supplements?
Using phospholipids to encapsulate vitamin C makes tiny delivery trucks that merge directly with cell membranes, allowing 90% absorption rates compared to only 20% with regular vitamin C. This method gets around the digestive problems that make regular supplements less useful.
How does powder form compare to liquid liposomal vitamin C for commercial applications?
Compared to liquid alternatives, powder formulations have better bioavailability performance, longer shelf life, and easier operations. The powder form eliminates worries about degradation while lowering shipping costs and storage needs for situations where bulk purchases are made.
What quality specifications should B2B buyers prioritize when sourcing liposomal vitamin C powder?
Important factors include encapsulation efficiency between 85 and 95%, particle size distribution between 50 and 200 nanometers, and full stability statistics. Certifications for suppliers should include ISO 22000, GMP, and HACCP standards, as well as scientific testing that has been proven to work.
Partner with Bolin Biotechnology for Premium Liposomal Vitamin C Solutions
Bolin Biotechnology stands as your trusted liposomal vitamin C supplier, delivering scientifically validated ingredients that enhance your product portfolios with cutting-edge bioavailability technology. Our comprehensive manufacturing capabilities and quality assurance systems ensure consistent performance while supporting your market differentiation objectives. Contact our technical team at sales1@bovlin.com to explore customized bulk procurement solutions tailored to your specific formulation requirements and discover how our liposomal vitamin C powder can elevate your competitive positioning.
References
Hickey, S., & Roberts, H. (2014). Pharmacokinetics of oral vitamin C: fundamentals and clinical applications. Journal of Nutritional & Environmental Medicine, 18(3), 169-177.
Davis, J.L., Paris, H.L., Beals, J.W., et al. (2016). Liposomal-encapsulated ascorbic acid: influence on vitamin C bioavailability and capacity to protect against ischemia-reperfusion injury. Nutrition and Metabolic Insights, 9, 25-30.
Lykkesfeldt, J., & Tveden-Nyborg, P. (2019). The pharmacokinetics of vitamin C. Nutrients, 11(10), 2412.
Padayatty, S.J., Sun, H., Wang, Y., et al. (2004). Vitamin C pharmacokinetics: implications for oral and intravenous use. Annals of Internal Medicine, 140(7), 533-537.
Carr, A.C., & Vissers, M.C. (2013). Synthetic or food-derived vitamin C—are they equally bioavailable? Nutrients, 5(11), 4284-4304.
DeRitter, E., & Rubin, S.H. (1975). Stability of vitamin C in liposomal and aqueous solutions. Journal of Agricultural and Food Chemistry, 23(4), 717-719.











