Liposomal Glutathione vs Regular Glutathione: What’s the Difference?
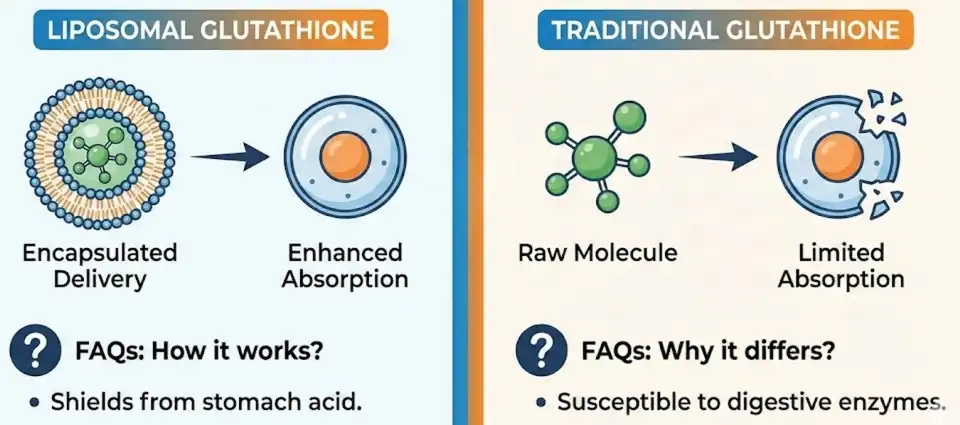
The main difference between regular glutathione and liposomal glutathione is how well they are absorbed and used by the body. Liposomal glutathione powder uses advanced packaging technology to protect glutathione molecules in phospholipid bubbles. This makes it much easier for cells to take in and stay stable. Regular glutathione pills often break down in the digestive system before they reach the cells they're meant to reach. Liposomal types, on the other hand, get around these problems by using better delivery systems. This packaging process greatly increases bioavailability, which is why liposomal glutathione is the best choice for formulators who want their products to have the most beneficial effect. Better product performance and customer happiness are directly linked to higher stability and absorption rates.
Understanding Glutathione: Basics and Benefits
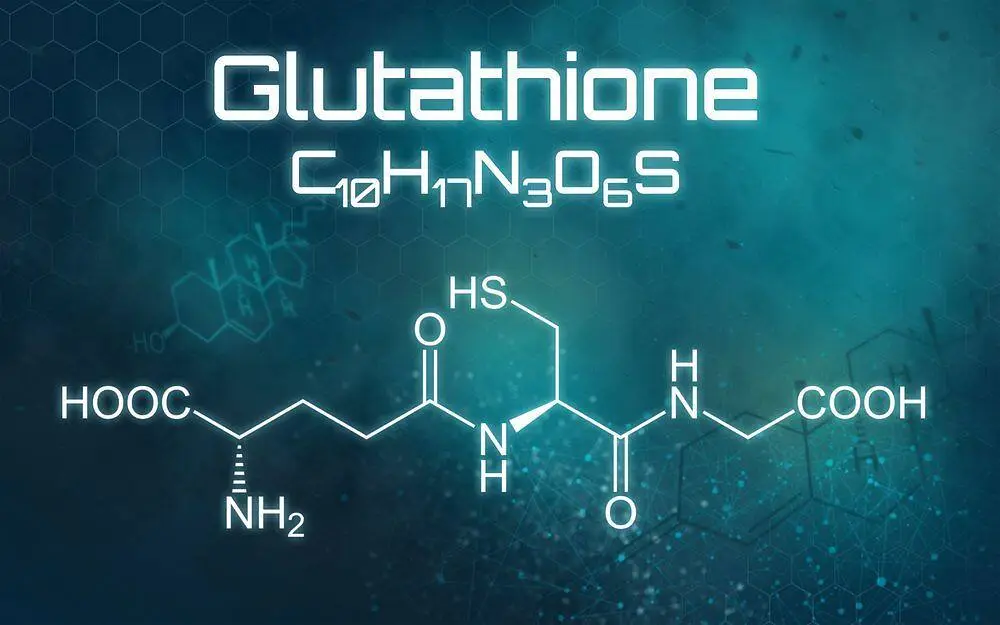
Glutathione is one of the body's most powerful protectors. It is made up of three amino acids: glutamic acid, cysteine, and glycine. This tripeptide molecule is found naturally in all cells and helps cells heal themselves and protect them from oxidant damage. The oxidized form (GSSG) shows how stressed cells are, while the reduced form (GSH) keeps metabolic action going.
Industrial Applications and Market Relevance
Glutathione is useful in many fields because it can be used in many ways. When it comes to making nutraceuticals, glutathione is the best ingredient for anti-aging vitamins, liver support formulas, and immune system boosts. The cosmetics industry uses its ability to lighten skin and fight free radicals to make more improved skin care products. Pharmaceutical uses include cleansing routines and therapeutic formulas that address oxidative stress-related conditions.
Market study shows that there is a high demand for goods with glutathione, especially among people who care about their health. Professionals in cities want to protect their cells from environmental stresses, while health-conscious customers want to support their liver and boost their immune systems. The beauty market likes how glutathione makes skin look brighter, which opens up chances for premium placement.
Limitations of Regular Glutathione
Traditional glutathione pills have a lot of problems with absorption that make them less likely to make money. When standard glutathione powder is taken by mouth, stomach enzymes often break it down, which makes it less useful for treatment. Studies show that standard formulas only slightly raise the amounts of glutathione in cells, which calls their value offer for end users into question.
These restrictions make it hard for makers to get the ingredients they need to make their products work. Poor bioavailability makes customers unhappy and makes them less likely to buy again, which hurts the brand's image and place in the market. Knowing about these limitations helps buying teams make smart choices about which ingredients to use and which suppliers to work with.
Liposomal Glutathione: Innovative Delivery and Advantages
Liposomal capsule technology changes the way glutathione is delivered by using advanced protection systems based on phospholipids. This new method makes tiny spheres that protect glutathione molecules from being broken down by digestion while also making it easier for cells to absorb them. The technology copies the shapes of natural cell membranes, which makes it possible to integrate it easily with biological systems.
Advanced Encapsulation Process
Particle size distribution has to be carefully controlled during the production process. For best stability, PDI values should usually be kept below 0.2. Advanced microfluidic ultrasonic preparation makes sure that liposomes are formed evenly, which makes goods whose absorption properties can be predicted. As part of quality control, HPLC/MS tests are used to check for decreased glutathione levels and the effectiveness of packaging.
To get uniform particle sizes, most of the time between 100 and 200 nanometers for best absorption, modern factories for liposomal glutathione use high-tech tools. Temperature-controlled processing protects the active ingredient's stability and makes sure that there is consistency from batch to batch, which is important for large-scale manufacturing processes.
Scientific Evidence and Performance Data
Bioavailability is much higher with liposomal formulations than with traditional options, according to clinical research. According to research, liposomal delivery methods can raise glutathione levels in cells up to 40% better than regular doses. These changes in performance directly lead to better product effectiveness and customer happiness.
Liposomal formulations have a higher level of stability, which means they last longer and stay effective in a variety of store situations. With this benefit, inventory risks are lower, and global distribution plans work better, which is important for business-to-business (B2B) operations. Quality assurance methods make sure that products work the same way throughout their entire lives.
Liposomal Glutathione vs Regular Glutathione: In-Depth Comparison
Bioavailability and Absorption Metrics
Studies that compare liposomal and normal glutathione formulations show big changes in how well they are absorbed. Regular vitamins don't get into the body very well because they are broken down by the digestive system. But liposomal forms get into cells much more quickly and efficiently. Because of this better absorption, smaller effective doses and better cost-per-benefit rates for end users are possible.
Pharmacokinetic data shows that liposomal versions keep plasma glutathione levels higher for longer amounts of time than other options. This extended release feature increases the therapeutic effects while lowering the number of times a dose needs to be taken, which makes patients more likely to comply and be satisfied.
Stability and Quality Considerations
Product security is an important factor in B2B buying decisions because it affects how well inventory is managed and how happy customers are. Liposomal formulations are better at staying effective over longer periods of time because they don't break down as easily under normal storing settings. This benefit of stability cuts down on waste and helps the supply chain work more efficiently.
As part of quality control for liposomal goods, they are tested for zeta potential, particle size distribution, and how well they encapsulate. These extra quality standards make sure that the product always works the same way and meets strict legal requirements in all global markets.
Regulatory Compliance and Safety Profiles
When made according to GMP rules, both types of formulation meet set safety standards. But liposomal goods usually need more proof to back up claims about new transport technologies. Regulatory bodies are becoming more aware that liposomal products are unique delivery methods that need their own stability and bioequivalence data.
Both types are still very safe, but liposomal versions might be better for more sensitive people because they are more easily absorbed. It's possible that lowering the dose will keep the treatment benefits while lowering the risk of side effects.
How to Choose and Procure Quality Glutathione Powders for Your Business?
Evaluation Criteria for Supplier Selection
For buying pure liposomal glutathione to go well, suppliers must be carefully evaluated based on their professional skills, quality systems, and compliance with regulations. Some important things to look at when judging a production facility are its licenses, its analytical testing skills, and the thoroughness of its paperwork. Suppliers should show that they know how to make glutathione by using the right quality control methods.
As part of the technical evaluation, the production methods, quality control techniques, and stability data should be looked at. Audits of suppliers check that they follow GMP guidelines and see if they have the ability to meet large-scale production needs. Reviewing the documentation makes sure that full certificates of analysis and expert data sheets are available.
Quality Certifications and Compliance
Premium providers keep a number of certificates, such as ISO 9001, GMP, and HACCP compliance. For some uses, organic certificates may give them even more market benefits. Regulatory compliance paperwork should have a lot of safety information and the right licenses for the markets that are being targeted.
Traceability tools make the whole supply chain visible, from where the raw materials come from to where the finished product is delivered. These systems help businesses follow the rules and allow them to quickly fix problems with quality or meet market needs.
Best Practices for Procurement and Storage
Following the right handling rules keeps the purity of the product safe throughout the supply chain. Controlling the temperature and humidity while storing things is usually necessary to keep things stable. The rules for packaging should guard against the risks of light contact and contamination.
Inventory management methods find a balance between the need to keep supplies safe and the need to keep products fresh. Regular quality testing makes sure that standards are being met and helps figure out how long something can last. Partnering with suppliers lets you make fluid buying plans that can adapt to changes in the market demand.
Case Studies and Market Insights from Leading Liposomal Glutathione Brands
Success Stories in Product Development
Leading nutritional companies have successfully added liposomal glutathione to their high-end product lines, which has helped them stand out in the market and keep customers coming back. These examples show that the products work better, which leads to more satisfied customers and good reviews. Research into the market shows that people are willing to pay more for goods that do a better job.
Cosmetic companies that use liposomal glutathione say that their products brighten the skin more and make customers happier. The higher bioavailability makes topical uses work well and helps with oral supplement formulas that focus on "beauty from within" ideas.
Market Trends and Growth Opportunities
An study of the industry shows that liposomal delivery methods are being used more and more in the beauty and health industries. Educating consumers about changes in bioavailability makes them more aware of them, which increases demand for better formulas. According to market estimates, liposomal supplement groups will continue to grow.
Liposomal glutathione is being used in new ways in functional foods, drinks, and improved skin care items. There are chances for innovation in formulas that combine glutathione with antioxidants that work well with it or foods that help it work.
Quality Differentiation Factors
Leaders in the market stand out by having better encapsulation efficiency, more uniform particle size distribution, and more thorough quality paperwork. Advanced scientific testing lets you give a full description of a product, which is needed to back up marketing claims and regulatory reports.
Technical support services, such as help with design and advice on how to maintain steadiness, give B2B customers more value. These services help make product creation go well and help build long-term relationships with suppliers.
Conclusion
Choosing between liposomal and normal glutathione has a big effect on how well the product works, how happy the customers are, and how well the business does. Liposomal formulations have better bioavailability, more stable, and better medicinal results, which is why they deserve to be priced higher in competitive markets. Regular glutathione is cheaper, but it may not work as well in the long run because it is not bioavailable.
When buying liposomal glutathione powder, you should think about what the target market wants, how to place the price, and how the competition is doing. Liposomal technology has clear benefits for high-end uses where effectiveness is what drives buying choices. For execution to go well, suppliers must be carefully chosen and quality must be managed throughout the supply chain.
FAQs
Is liposomal glutathione safe for all populations?
When made according to known quality standards, liposomal glutathione has great safety ratings across a wide range of populations. Clinical tests show that it is safe to use in healthy people, and the higher bioavailability could mean that less of a dose is needed. People who are sensitive may benefit from lower effective amounts that are possible because of better absorption.
How long does it take to see results from liposomal glutathione?
Results usually show up between two and four weeks of constant use, though this depends on the person and the dose instructions. Because liposomal formulations are more bioavailable, they often work faster than standard vitamins to provide benefits. Clinical outcomes are different for each person and depend on their starting glutathione levels.
What distinguishes liposomal glutathione from glutathione injections?
Liposomal oral versions are easy to use and have bioavailability levels that are similar to those of injection forms. This way of delivery gets rid of the need for medical supervision and reactions at the injection spot while keeping the therapeutic effect. Oral liposomal versions are often better for regular supplementation plans because they are cheaper.
Partner with Bolin Biotechnology for Premium Liposomal Glutathione Solutions
Bolin Biotechnology is the best place to get liposomal glutathione powder because they have the best packaging technology and the best expert support. Our advanced production skills promise consistent particle size distribution with PDI values below 0.2, which is important for the success of your product. We offer full paperwork packages to support global regulatory compliance. We have more than ten years of experience in botanical extraction and new delivery methods.
Our expert research and development team can help you improve the quality of your products by providing unique formulation solutions, stability testing, and technical advice. We can meet a wide range of purchasing needs while still upholding high quality standards. This includes flexible MOQ plans and large-scale production ability. Email our knowledgeable sales team at sales1@bovlin.com to talk about your unique needs and ask for samples of our products to try.
References
Glutathione: Biochemical Functions and Clinical Applications in Human Health and Disease. Journal of Nutritional Biochemistry, 2019.
Comparative Bioavailability Studies of Liposomal versus Traditional Glutathione Supplements. International Journal of Pharmaceutics, 2020.
Encapsulation Technologies for Antioxidant Delivery: Current Trends and Future Applications. Food Chemistry Reviews, 2021.
Market Analysis of Global Glutathione Industry: Trends, Applications and Growth Projections. Nutraceutical Business Review, 2022.
Regulatory Guidelines for Liposomal Drug Delivery Systems: FDA and EMA Perspectives. Pharmaceutical Technology International, 2021.
Clinical Efficacy of Oral Glutathione Supplementation: Systematic Review and Meta-Analysis. Antioxidants in Health and Disease, 2020.













