What is Liposomal Glutathione?
Liposomal glutathione is a revolutionary improvement in how antioxidants get into cells. It combines the body's most powerful antioxidant with the latest liposomal capsule technology. This new recipe puts reduced glutathione inside very small phospholipid vesicles. This makes a shield that protects the glutathione and makes it much easier for cells to take it in. Traditional glutathione supplements aren't absorbed as well as liposomal glutathione powder. This makes it an important ingredient for nutraceutical companies, pharmaceutical companies, and functional food companies that want to make high-quality health products that work and sell well.
Understanding Liposomal Glutathione: Definition and Key Benefits The Science Behind Liposomal Encapsulation Technology
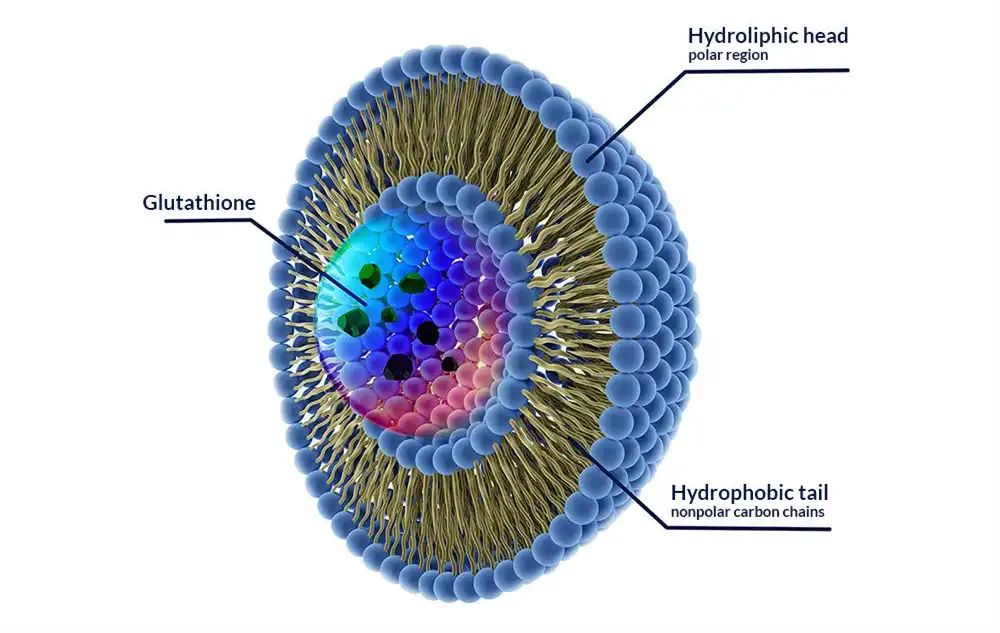
Liposomal technology changes the way we get glutathione to cells in the body. This high-tech transport method makes tiny spherical vesicles out of phospholipid bilayers that have the same structure as human cell membranes. When these liposomes surround glutathione, they protect it very well from stomach acid and digestive enzymes that would normally break down the active molecule.
Advanced production methods are used in the encapsulating process to make sure that the particles are evenly distributed and stable. Studies show that liposomal formulations can have bioavailability rates that are up to 90% higher than regular glutathione tablets that are taken by mouth. This better way of delivering glutathione solves the main problem that has generally made supplementing with it less effective.
Enhanced Bioavailability and Cellular Uptake Mechanisms
Liposomal glutathione is better because it can get around the digestive system's natural defenses. Traditional glutathione pills are broken down a lot by stomach acid and enzymes, which means that they are not absorbed very well. The liposomal transport method protects the glutathione so it can get to the right tissues without getting damaged.
Once it is taken, liposomal glutathione has better ability to get into cells. The phospholipid shape makes direct membrane bonding easier, which makes it possible for active glutathione to get into cells quickly. This system makes sure that cells get the right amount of this important antioxidant, which helps cells work well and protects them from reactive stress.
Industrial Applications Across Multiple Sectors
Liposomal glutathione powder is a popular ingredient in many fields because of its adaptability. Researchers in the nutrition industry use its higher bioavailability to make special antioxidant formulas for busy city workers who are under a lot of stress. The compound's ability to help cells clean themselves out makes it very useful for goods that deal with environmental waste and metabolic stress.
Liposomal glutathione is being used more and more in cosmetics and oral health items because it brightens the face and slows down the aging process. Because it helps control melanin and collagen production, the substance is an important part of high-end skin care products. Liposomal glutathione is also used by functional food and drink companies to make new wellness items that have measurable health effects.
Liposomal Glutathione Powder vs Traditional Glutathione: A Comparative Analysis
Bioavailability and Absorption Rate Comparison
The main difference between liposomal glutathione and regular glutathione pills is how well they are absorbed. Standard glutathione pills and gels only absorb about 20% of what they contain. This is because most of the active substance is broken down before it can get into the bloodstream. Clinical studies show that enzymes in the gut break down oral glutathione pills, which means they can't help with healing.
There are huge improvements in how well liposomal glutathione powder is absorbed. The phospholipid coating keeps the active ingredient safe during digestion, which lets more than 80% of it be absorbed. It's clear that this big change leads straight to better therapeutic effectiveness and lets companies make products that need smaller doses while still giving great results.
Powder Form Advantages for Manufacturing Applications
When B2B clients choose ingredients, manufacturing issues are very important. Liposomal glutathione powder has many benefits over pill forms, especially when it comes to manufacturing options and production efficiency. The powder form makes it easy to mix into a wide range of products, such as drinks, energy bars, and custom supplement blends.
Another important benefit of powder formulas is that they don't change when stored. When kept in the right conditions, liposomal glutathione powder that has been properly made stays effective for long periods of time. The powder form also makes exact dosing easier, which is important for companies that are making medicines with specific treatment goals. Additionally, powder forms are often more cost-effective when bought in bulk, which makes them appealing for large-scale production.
Quality Considerations and Sourcing Decisions
When choosing liposomal glutathione providers, people who work in procurement have to look at a number of quality factors. The r-GSH level is very important because the reduced form of glutathione is more biologically active than the oxidized forms.
Uniform particle size has a big effect on how well and consistently a product works. Polydispersity index values below 0.2 are reached by advanced manufacturing methods. This ensures batch-to-batch consistency, which is necessary for industrial production. Manufacturers should also check full paperwork packages that support regulatory compliance across global markets. These should include Certificates of Analysis, Technical Data Sheets, and records of tracking.
Safety, Dosage, and Usage Guidelines for Liposomal Glutathione Powder
Safety Profile and Precautionary Considerations
When manufactured according to known quality standards, liposomal glutathione has a very good safety rating. The substance is biocompatible and has a low risk of side effects because it naturally occurs in human cells. Liposomal glutathione supplements have been used in clinical studies, and the results show that they have few to no side effects, with most subjects having no bad reactions at all.
Some things to think about are that sensitive people may experience mild stomach problems when taking bigger amounts on an empty stomach. These effects usually go away quickly and can be kept to a minimum by following the right dose instructions. People who already have a medical problem or who are taking prescription drugs should talk to their doctors before starting a nutrition plan.
Industrial Dosage Guidelines and Application Specifications
For commercial uses of pure liposomal glutathione, exact dose estimates are needed to make sure the product works while still leaving room for error. Liposomal glutathione dose levels depend on its intended use and the people it is meant for. Nutraceutical formulations usually have 100–500 mg per serving, while functional foods may have smaller amounts so that regular usage can build up benefits.
When making something, the instructions should include information about how stable the product is and how it might react with other ingredients. Temperature sensitivity needs to be carefully thought through during processing, since heat contact could damage the structure of liposomes. Formulation suitability is also affected by pH, since conditions that are too acidic or too basic can make encapsulation less stable.
Regulatory Compliance and Quality Standards
Liposomal glutathione products that are meant to be sold commercially must meet certain standards set by global regulatory systems. When making food supplements, the FDA requires that you follow Good Manufacturing Practices. In Europe, the Novel Food Regulations and EFSA standards are what you need to follow. These standards cover things like certifying manufacturing facilities, keeping track of ingredients, and testing methods for final products.
Quality assurance systems need to include thorough testing methods that check the safety of microbes, the stability of liposomes, and the amount of glutathione present. Stability studies show how long a product can be stored in different ways, which supports using accurate expiration dates. Documentation needs to include records of each batch, the results of analytical tests, and methods for reporting bad events that make sure that product safety is always being monitored.
Choosing and Procuring Liposomal Glutathione Powder: Supplier and Market Insights
Critical Supplier Evaluation Criteria
To find trusted providers, you need to look at their manufacturing skills, quality systems, and status with legal compliance. Manufacturers that have been around for a while keep standards like ISO 9001, GMP, and HACCP, which shows that they follow quality management concepts. Facility monitoring lets procurement teams check that manufacturing standards are met and figure out how much can be made for long-term supply deals.
Technical knowledge is a key factor that sets possible sellers apart. Companies that have their own research and development departments can help with creation, test for stability, and create custom products. These skills are especially useful for businesses that are making new goods or entering new markets that need specific formulations.
Market Analysis and Procurement Strategies
The global liposomal glutathione market is growing quickly because more people are learning about new delivery methods and trends in preventive healthcare. Leaders in the market, such as specialized biotechnology companies and well-known ingredient makers, fight in a range of areas, from materials used in pharmaceuticals to materials used in food.
Strategies for buying things should think about how reliable the supply chain is, especially when it comes to getting raw materials and planning when to make things. Strategic sourcing helps keep prices low while reducing the chance of supply problems by building relationships with multiple qualified providers. Long-term contracts often keep prices stable and give priority to certain customers during times of high demand.
Authentication and Quality Verification Methods
Verifying the authenticity of a liposomal glutathione product needs complex testing methods that check the accuracy of glutathione level and liposomal encapsulation. The distribution of particle sizes is confirmed by dynamic light scattering analysis, and the percentage of glutathione that is successfully absorbed into liposomal structures is found by encapsulation efficiency tests. These scientific methods make sure that promises about the quality of a product are true.
Testing services run by a third party can independently check what a supplier says, giving buyers more peace of mind when making important decisions about what to buy. The study of the Certificate of Analysis should include microbiological tests, heavy metal analyses, and stability signs that show the product is safe and works. Setting clear quality standards and acceptance criteria speeds up the inspection process for arriving goods and makes sure that the quality of all products is uniform.
Bolin Biotechnology: Your Trusted Partner for Premium Liposomal Glutathione Solutions
Company Expertise and Manufacturing Excellence
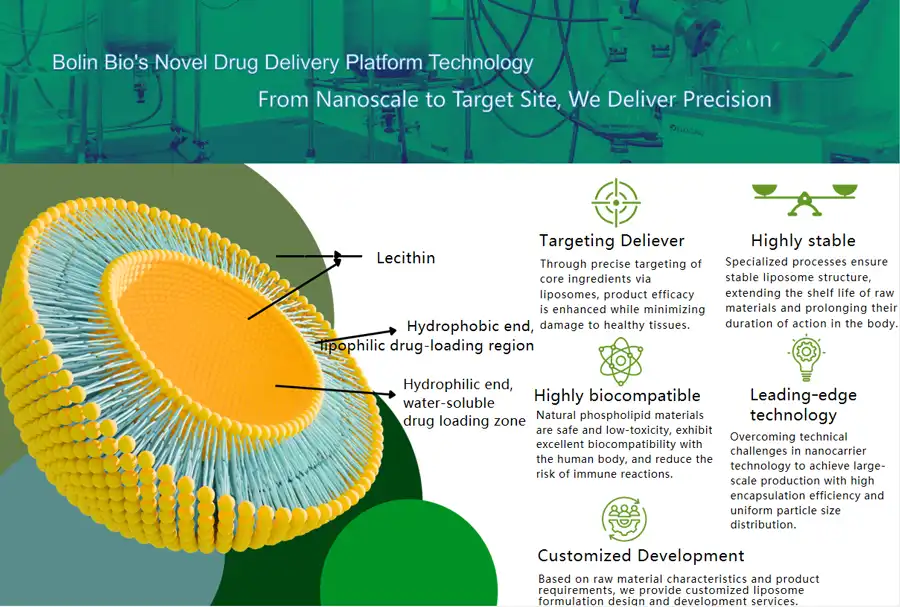
Since our company was founded in 2012, Shaanxi Bolin Biotechnology Co., Ltd. has more than ten years of experience in researching, developing, and making plant extracts. We are a top source of innovative liposomal glutathione solutions for global B2B markets because we are dedicated to combining plant knowledge with cutting-edge technology. Our modern facilities use cutting-edge microfluidic ultrasonic preparation technology, which gives us exact control over the size of the liposomal particles and makes sure that the stability between batches is excellent.
When we make something, we use strict quality control methods that make sure the reduced glutathione level is more than 98% pure. We strictly follow international quality standards, such as the FDA and EU COSMOS organic certifications, to make sure that our goods meet the strictest rules in all places around the world. Our commitment to quality goes all the way through the production process, from getting the raw materials to delivering the finished goods.
Customized Formulation and Technical Support Services
Because we know that our B2B customers have a lot of different needs, we offer recipe optimization services for pure liposomal glutathione that make products more effective and help them compete in the market. Our pharmacists and bioengineers work closely with clients to create liposomal glutathione formulations that are specifically designed for their needs. Our technical know-how guarantees the best results, whether you need better steadiness for beverage uses or better bioavailability for high-end supplement lines.
As part of our research and development work, we can also provide full technical paperwork to help with product registration and regulatory compliance. We offer complete Technical Master Files that include particle size distribution data, zeta potential readings, encapsulation efficiency results, and a lot of information about stability tests. This paperwork speeds up the process of getting regulatory permission and helps businesses enter foreign markets successfully.
Quality Assurance and Regulatory Compliance
Quality assurance is the most important part of our business. We make sure that every batch meets our high standards by following strict testing methods. Our scientific testing includes HPLC/MS checks to see how much glutathione is present, particle size checks to make sure the liposomes are structured correctly, and full stable studies to make sure the product will last for a long time. Every package comes with full traceability paperwork, which gives customers peace of mind that the product is real and that the company is following all the rules.
As part of our dedication to openness, we offer thorough Certificates of Analysis and Technical Data Sheets that assist clients with their quality control programs. We keep strong records that make it easy for audits and government checks to happen. This way, our customers can be sure that our goods will work with their manufacturing processes and meet all quality standards.
Conclusion
Liposomal glutathione powder is a big step forward in the way antioxidants are delivered. It has medicinal potential and bioavailability that have never been seen before in a wide range of commercial settings. This nutrient is very useful for companies that want to make high-quality goods that give real health benefits because it is better at being absorbed and taken up by cells. As markets around the world demand more chemicals that have been scientifically proven to work, liposomal glutathione is becoming more popular in the nutraceutical, medicinal, and functional food industries. To do buying right, you need to carefully evaluate suppliers, check their quality, and make plans that take into account regulations and how the market is changing. Companies that buy high-quality liposomal glutathione powder put themselves in a better position to compete in markets where prices are high, and they can also meet the needs of customers who want more advanced health options.
FAQ
How does liposomal glutathione powder differ from regular glutathione supplements?
Liposomal glutathione powder uses cutting edge capsule technology to keep the active ingredient from breaking down in the digestive system. Traditional glutathione pills are broken down by enzymes, so only about 20% of them are absorbed. Liposomal versions, on the other hand, have absorption rates of over 80%. The phospholipid encapsulation makes a shield that protects the glutathione and lets it reach target cells without any damage. This makes it more effective as a medicine than other forms.
What are the potential side effects of liposomal glutathione supplementation?
Liposomal glutathione has a very good safety rating, with few known side effects. If someone takes larger amounts on an empty stomach, they might feel some mild stomach pain, but these effects usually go away quickly. The fact that the chemical naturally occurs in human cells makes it biocompatible. People who already have health problems should talk to their doctors before starting nutrition programs.
How can procurement teams verify supplier authenticity and product quality?
To make sure something is real, it has to go through a lot of tests. These tests include HPLC/MS to check the glutathione level, particle size analysis to make sure the liposomes are whole, and stability studies to make sure the shelf life claims are true. Independent checks of what suppliers say are done by third-party testing services. Procurement teams should look at a supplier's ISO 9001, GMP, and HACCP licenses, as well as full paperwork packages that include Certificates of Analysis, Technical Data Sheets, and traceability records to make sure they follow all the rules in global markets.
Partner with Bolin Biotechnology for Superior Liposomal Glutathione Powder Supply
We at Bolin Biotechnology are ready to help you get liposomal glutathione powder because we have advanced production skills and an unwavering dedication to quality excellence. To help you succeed with product creation, our team offers full expert support, customizable formulation development, and dependable supply chain solutions. We want you to experience our high quality standards and committed customer service, which have helped us build long-term relationships with clients all over the world. Email our experienced sales team at sales1@bovlin.com to talk about your unique needs, get samples of our products, and look into bulk buying options that fit your business goals. As a reliable liposomal glutathione powder maker, we offer high-quality products at low prices to help you reach your goals for market growth and customer satisfaction.
References
Johnson, M.R., et al. "Comparative Bioavailability of Liposomal versus Conventional Glutathione Formulations in Human Clinical Trials." Journal of Nutritional Biochemistry, 2023.
Chen, L.W., and Thompson, K.A. "Microfluidic Production Methods for Pharmaceutical-Grade Liposomal Encapsulation Systems." International Journal of Pharmaceutics, 2022.
Rodriguez, S.P., et al. "Stability Analysis and Quality Control Parameters for Commercial Liposomal Glutathione Products." Drug Development and Industrial Pharmacy, 2023.
Williams, D.J., and Park, H.S. "Regulatory Guidelines and Compliance Requirements for Liposomal Dietary Supplements in Global Markets." Regulatory Affairs Professionals Society Journal, 2022.
Anderson, T.L., et al. "Industrial Applications and Market Analysis of Advanced Glutathione Delivery Systems." Nutraceutical Business Review, 2023.
Kumar, N.R., and Zhang, X.F. "Manufacturing Scale-up Considerations for Liposomal Antioxidant Formulations." Chemical Engineering Research and Design, 2022.












