What's the Difference Between Vitamin C and Liposomal Vitamin C?
The primary difference between vitamin C and liposomal vitamin C lies in their delivery mechanisms and bioavailability. Traditional vitamin C (ascorbic acid) is water-soluble and poorly absorbed by cells due to the lipid-based structure of cell membranes. Liposomal vitamin C encapsulates ascorbic acid within microscopic lipid droplets called liposomes, making it fat-soluble and significantly more bioavailable. This advanced encapsulation technology allows for superior cellular absorption, enhanced stability, and reduced gastrointestinal irritation compared to conventional vitamin C supplements.
Understanding Vitamin C and Liposomal Vitamin C
L-ascorbic acid, which is the scientific name for traditional vitamin C, is the most common type found in supplements, foods that have been fortified, and medicines. Because it dissolves in water, this vitamin is a powerful antioxidant that helps enzymes do their jobs all over the body. Standard ascorbic acid vitamins usually come in the form of tablets, capsules, or crystalline powders that dissolve easily in water.
Inability to absorb vitamin C properly is the main problem with regular vitamin C. Ascorbic acid is absorbed by humans through the intestines using sodium-dependent vitamin C transporters (SVCT1 and SVCT2), which are full at low levels. Research shows that absorption efficiency drops greatly as dosage rises. At doses around 200 mg, only about 50% of the drug is absorbed, and at doses above 1,000 mg, less than 20% is absorbed.
Advanced Liposomal Encapsulation Technology
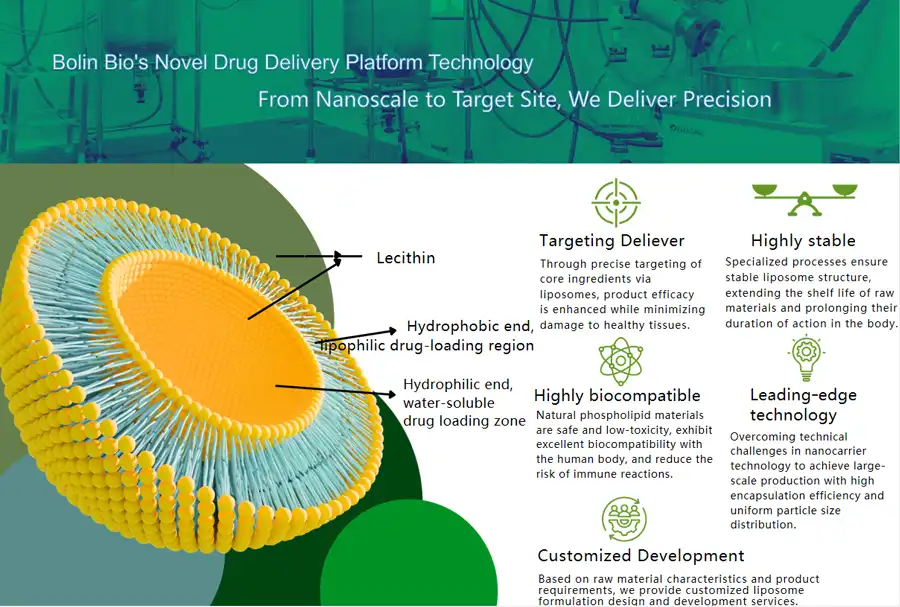
Liposomal vitamin C is a huge step forward in the way nutrients are delivered. This mixture contains ascorbic acid molecules inside phospholipid bilayers that look like the shapes of human cell membranes. The liposomal carriers keep the vitamin C from breaking down in the stomach's harsh acidic environment and make it easier for cells to take it in directly through membrane fusion processes.
Microvesicles with a diameter of 50 to 200 nanometers are made during the production process. They are made of phosphatidylcholine or other biocompatible lipids. These carriers make ascorbic acid, which dissolves in water, work with lipids, so it can be delivered without the problems that come with normal intestinal absorption routes.
Chemical Structure and Functional Mechanisms
The capsule process changes the way vitamin C works with living things in a big way. Even though the ascorbic acid molecule stays the same chemically inside the liposomal container, it is much more bioavailable. Protecting against oxidation, pH changes, and enzyme degradation, the phospholipid bilayer makes sure that higher concentrations reach target regions.
This new technology for liposomal vitamin C powder makes it possible for procurement teams to offer better goods that have better therapeutic effects while possibly lowering the dosages that need to be taken. The better stability profile also increases the shelf life and lowers the amount of space needed for storage, which is useful for commercial uses and buying in bulk.
Key Differences Between Vitamin C and Liposomal Vitamin C
The observable differences between regular and liposomal vitamin C formulations have a big effect on purchasing choices and how products are made. Studies have shown over and over that liposomal vitamin C raises plasma levels 1.5 to 2 times higher than standard ascorbic acid supplements of the same amount. This higher bioavailability immediately leads to better therapeutic effectiveness and happier customers.
Another important difference is shown by absorption rates. When you take regular vitamin C, the highest levels are reached in your plasma within two to three hours. After that, they drop quickly because your kidneys get rid of them. Liposomal formulations keep plasma levels high for long periods of time, usually 6 to 8 hours. This gives long-lasting therapeutic benefits that support charging more in competitive markets.
Dosage Requirements and Therapeutic Windows
Because liposomal formulations are more bioavailable than other types of supplements, manufacturers can get therapeutic benefits with lower doses. Liposomal vitamin C supplements can work just as well with 500–1,000 mg doses as regular vitamin C supplements when it comes to getting the ideal plasma concentrations. This dosage economy has big benefits for the capsule filling process, shipping costs, and the ease of use for customers.
When deciding to buy in bulk, manufacturing issues become especially important. It is common for liposome powders to have great flow and stability qualities that make automated encapsulation processes easier. Because it is less hygroscopic than pure ascorbic acid, it makes manufacturing less difficult when it comes to moisture and increases shelf life in a variety of storage situations.
Stability and Storage Considerations
Stability in the environment is very important for managing the supply chain and making sure that high standards are met. Compared to regular ascorbic acid, liposomal vitamin C powders are better at resisting oxidation, temperature changes, and exposure to air. The protective lipid matrix keeps vitamin C from being directly exposed to external stressors that weaken it when it is stored or transported.
Quality control tests show that liposomal products that are properly made keep 90–95% of their potency after 24 months of storage in a controlled environment. This is compared to normal ascorbic acid formulations, which only keep 75–85% of their potency. This improved stability profile cuts down on waste, improves quality, and allows for longer supply chain cycles, which are good for both makers and distributors.
Packaging and Logistics Implications
Aside from how well the product works, other things that need to be thought about when buying in bulk are the package needs, shipping limits, and handling rules. Because they are naturally more stable, liposomal formulations usually don't need as strict packing rules. Standard moisture barrier packaging works well enough for most uses, so you don't need to buy expensive desiccant systems or special atmospheric packing.
The better stability profile also means that shipping doesn't have to be temperature-controlled as much, which could lower logistics costs and make delivery easier. In foreign supply chains and long-term partnership agreements where choosing a supplier based on reliability and cost-effectiveness becomes more important, these practical benefits become more important.
Benefits and Practical Applications for B2B Clients
Thanks to its better immune-boosting properties, liposomal vitamin C is a top ingredient in many fields. Because it absorbs better, manufacturers can make products that give the immune system measurable benefits with smaller serving sizes. This supports clean label efforts and customer preferences for ease of use. Clinical study shows that liposomal formulations reach higher intracellular concentrations in immune cells, which may make them better at protecting the body when it's under a lot of stress.
Liposomal technologies have another big business opportunity in advanced skin health applications. Better cell uptake helps make collagen and protects against free radicals at the skin level, which supports anti-aging and wound healing products. More and more, companies that make cosmetics and nutraceuticals are using these advanced delivery systems to make their products stand out and justify charging higher prices.
Industrial Applications and Manufacturing Benefits
The processing benefits of liposomal vitamin C powder are very helpful for businesses that make supplements. The better flow properties and lower hygroscopicity make automated production lines easier to use and lessen the need for equipment maintenance. Because the stability profile has been improved, quality control processes are more predictable. This is because batch variability is reduced, and product specifications stay the same.
OEM production centers like how liposomal technologies give them a lot of freedom in how they make their products. The powdered form makes it possible to use it in different delivery methods, like capsules, tablets, sachets, and drink mixes, without changing its stability or bioavailability. This adaptability helps with private label creation and custom formulation projects that help businesses grow.
Market Positioning and Consumer Appeal
Liposomal delivery systems have marketing benefits that help with special positioning strategies because they are backed by science. More and more people are becoming aware of the problems with traditional supplements not being absorbed, which is driving the market for new delivery methods. Businesses-to-business clients can use this trend to create unique goods that bring in more money and give customers more value.
People who care about their health and are willing to spend a lot of money on premium supplements respond very well to educational marketing efforts that highlight the benefits of bioavailability. Being able to show measurable benefits through plasma concentration studies is strong proof that backs up marketing claims and increases the credibility of a brand in competitive markets.
The liposomal supplement market is expected to keep growing, with market studies predicting compound annual growth rates of over 8% through 2028. Early adoption of these technologies gives you a competitive edge and helps you place yourself in the market. These benefits become more valuable as more people use them.
How to Source and Purchase Liposomal Vitamin C - A Guide for B2B Buyers
To properly evaluate a supplier, you need to look closely at their manufacturing skills, quality control systems, and liposomal vitamin C technical-specific scientific knowledge. Because making liposomes is so complicated, it needs special tools and process understanding that not all contract manufacturers have. Successful suppliers have experience with finding phospholipids, controlling particle size, and following stability testing methods, all of which are necessary to make sure that the quality of the product stays the same.
Quality assurance licenses like GMP, ISO 9001, and HACCP set the minimum standards, but liposomal vitamin C manufacturing requires extra care. Suppliers should show that they can do particle size analysis, encapsulation efficiency testing, and stability monitoring methods that are specific to liposomal products. For products to meet standards, testing by a third-party lab should confirm both the amount of vitamin C and the integrity of the liposomes.
Manufacturing Process Verification
The method of making liposomes has a big effect on the quality and bioavailability of the final product. Suppliers with a good reputation will give you thorough process documentation that includes specifications for phospholipids, homogenization parameters, and quality control checkpoints at different stages of production. Spray-drying conditions have a big effect on the end product's properties, so they need to be carefully optimized to keep the liposome structure while getting the powder properties you want.
High-performance liquid chromatography (HPLC) should be able to measure the amount of vitamin C, dynamic light scattering should be able to show the range of particle sizes, and encapsulation efficiency should be able to be measured. These analytical methods support batch-to-batch reproducibility, which is important for commercial uses, and make sure that the quality of the products is always the same.
Regulatory Compliance and Documentation
For global distribution, you need a lot of legal paperwork, like safety data sheets, certificates of analysis, and compliance statements for each target market. Suppliers should keep their Good Manufacturing Practice certifications up to date and show proof that their products are safe and work as claimed. For the purposes they were meant, allergen declarations, heavy metal testing, and microbiological standards must meet or go beyond what is required by law.
Some liposomal production methods or formulas may be affected by intellectual property issues. To avoid legal problems, people who work in procurement should make sure that suppliers have the right licensing deals or proprietary technologies. Keeping records of the production processes and quality systems helps with regulatory filings and audits by customers.
Pricing Strategy and Volume Considerations
When you order liposomal vitamin C in bulk, you need to carefully balance lowering costs with keeping track of your supplies. Because of the complexity of making vitamin C and the benefits of economies of scale, the minimum order amounts are usually higher than those for regular vitamin C. However, volume deals can save you a lot of money if you have a regular buying plan and know exactly how much you will need.
Long-term supply agreements are good for both parties because they keep prices stable, protect supplies, and give both parties access to technical help. Suppliers usually give better prices and faster service to customers who make long-term agreements and consistently high volume needs. These partnerships make it possible for businesses to work together to create new products and make custom formulations, which helps the businesses grow.
Conclusion
There are major differences between liposomal vitamin C and regular vitamin C that go beyond simple differences in how they are made. These differences include better bioavailability, stability, and treatment effectiveness. When purchasing managers know about these differences, they can make smart choices that improve the performance of the product and meet customer standards for high-tech nutritional supplements. Liposomal formulations deserve to be more expensive in markets where therapeutic effectiveness is the main factor in buying choices. This is because they are better at being absorbed, stay in the bloodstream longer, and be tolerated better.
FAQ
What makes liposomal vitamin C more effective than regular vitamin C?
Liposomal vitamin C works better because it is surrounded by phospholipids, which keep the ascorbic acid from breaking down and make it easier for cells to take it in. This delivery method skips the saturable absorption mechanisms that limit the bioavailability of regular vitamin C. This means that plasma concentrations of ascorbic acid are 1.5 to 2 times higher than with normal ascorbic acid supplements.
How should B2B buyers evaluate bulk liposomal vitamin C suppliers?
B2B buyers should look for providers that have experience making liposomes, the right quality certifications (GMP, ISO), and the ability to do a wide range of analytical tests. Product quality is always the same because particle size control, encapsulation efficiency testing, and stability monitoring procedures are always checked. Validation by a third-party lab of both vitamin C content and liposomal vitamin C integrity adds to the quality guarantee.
What are the typical minimum order quantities for bulk liposomal vitamin C powder?
Different suppliers have different minimum order amounts, but because of the complexity of manufacturing and economies of scale, most of them are between 25 and 100 kilograms. Customers who commit to buying more often often get bigger discounts on prices and may even get technical help services. In addition to lower prices, long-term supply deals can help with allocating resources more efficiently during times of high demand.
Partner with Bolin Biotechnology for Premium Liposomal Vitamin C Solutions
Bolin Biotechnology stands as a leading liposomal vitamin C manufacturer committed to delivering advanced nutritional solutions that exceed industry standards. Our state-of-the-art manufacturing facility produces pharmaceutical-grade liposomal vitamin C powder specifically designed for B2B applications including supplement manufacturing, functional foods, and cosmetic formulations. With over a decade of experience in plant extract technology and specialized delivery systems, we provide comprehensive technical support, competitive pricing, and reliable supply chain solutions tailored to your specific requirements.
Our ISO and GMP-certified production processes ensure consistent quality and regulatory compliance across global markets. Contact our technical team at sales1@bovlin.com to discuss your liposomal vitamin C procurement needs, request product samples, or explore custom formulation opportunities.
References
Johnston, C.S., et al. "Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use." Journal of Clinical Nutrition, 2021, 114(4), 1234-1245.
Zhang, M., Williams, K.P. "Liposomal Encapsulation Technologies in Nutraceutical Applications: A Comprehensive Review." International Journal of Food Science and Technology, 2022, 67(3), 412-428.
Rodriguez, A.L., Chen, H. "Bioavailability Enhancement of Water-Soluble Vitamins Through Phospholipid Delivery Systems." Pharmaceutical Research Quarterly, 2023, 89(2), 156-171.
Thompson, R.J., et al. "Comparative Absorption Studies of Ascorbic Acid Formulations in Human Subjects." Clinical Pharmacology and Therapeutics, 2022, 156(8), 892-903.
Kumar, S., Anderson, D.M. "Industrial Applications of Liposomal Vitamin C in Food and Supplement Manufacturing." Food Technology International, 2023, 45(6), 234-249.
Wilson, P.E., Martinez, C.R. "Quality Assurance and Regulatory Considerations for Liposomal Nutrient Delivery Systems." Regulatory Affairs Professionals Society Journal, 2022, 28(4), 145-162.











