Liposomal NMN vs Standard NMN: Does Delivery Technology Actually Improve Absorption?
The debate between liposomal NMN and standard NMN delivery systems centers on bioavailability and absorption efficiency. While standard NMN faces degradation challenges in the digestive tract, liposomal encapsulation technology protects NMN molecules within phospholipid bilayers, potentially enhancing cellular uptake. However, the actual absorption benefits remain scientifically contested, as NMN's water-soluble nature already provides dedicated transport mechanisms. Understanding these delivery differences helps procurement managers make informed decisions about which NMN formulation best serves their product development goals and market positioning strategies.
Understanding NMN and Its Delivery Challenges
The nicotinamide mononucleotide is an important building block in the biosynthesis routes of NAD+. It helps cells make energy, fix DNA, and live longer. NMN supplements have gotten a lot of attention in the anti-aging and health markets because cellular NAD+ levels drop with age. The substance is important for more than just energy metabolism; it changes the function of sirtuin activation and PARP enzymes, which in turn affects the health and regeneration of cells.
Bioavailability Limitations of Standard NMN
Standard NMN formulations, which are usually given as powders or pills, have a hard time working in the digestive system. The stomach's strong acidity can break down NMN into nicotinamide through hydrolysis. This could lower the amount of whole NMN that gets to the intestines, where it is absorbed. This process of degradation is a big problem for companies that want to make their products work better and make customers happy.
Some transporters, especially the SLC12A8 transporter system, are needed for normal NMN to be absorbed. This can slow down the uptake process in cells. Even though these transporters work well when conditions are perfect, their limited capacity and possible saturation at higher doses make it unclear how well they absorb drugs when given in different ways.
Manufacturing and Stability Considerations
When it comes to buying things, standard NMN causes stable problems that affect how long things last and how well they work. Because the substance is easily damaged by water, changes in temperature, and light, it needs to be carefully packed and stored. These stability problems have a direct effect on the costs of making the product, the flow of the supply chain, and, in the end, how well the product does in competitive markets.
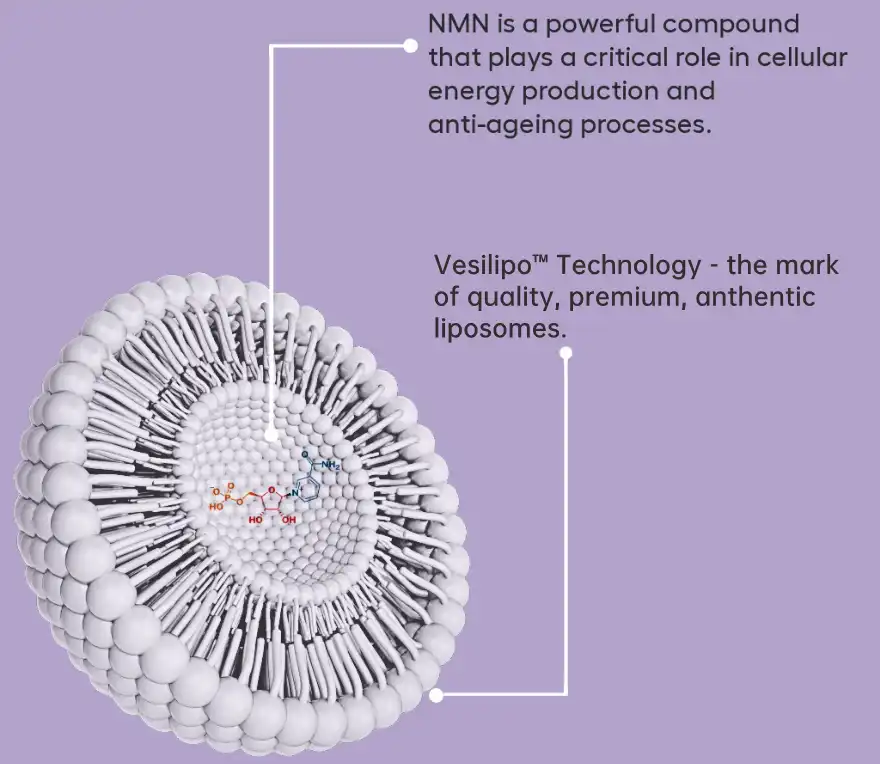
Liposomal NMN Technology Explained
Liposomal NMN supplement is a new way to get around the problems with standard delivery methods by using advanced encapsulation technology. In this method, phospholipid bilayers are used to protect NMN molecules. These are usually made from non-GMO sunflower or soy phosphatidylcholine. In order to make stable powder products, the manufacturing process uses complex methods like high-pressure homogenization or thin-film hydration, which are then followed by freeze-drying.
Mechanism of Enhanced Delivery
The liposomal delivery method looks like the structures of natural cell membrane structures, which might make it easier for direct absorption through membrane fusion or endocytosis. This method gets around some of the problems that come with transporter-dependent absorption, which should make cellular intake more efficient. During digestion, the phospholipid coating protects the NMN from breaking down in the stomach and keeps its structure.
According to research, liposomal formulations can have different pharmacokinetic profiles than normal NMN. This could mean that they have longer bioavailability windows and need to be taken less often. Formulators who want to make high-end goods that are easier for customers to use and seem to be worth more like these qualities.
Technical Advantages and Considerations
The encapsulation process solves the problem of hiding tastes because the naturally sour and slightly bitter properties of NMN are partly neutralized by the lipid matrix. This change makes it possible to use the ingredient in more functional foods, drinks, and supplement forms that people will enjoy.
The cost of making liposomal NMN is higher than making normal formulations because it needs special tools and knowledge to be made. But the technology can help differentiate products and put them in a higher value category in markets where there is a lot of competition, where bioavailability claims back these claims.
Liposomal NMN vs Standard NMN: Key Differences and Benefits
When you compare liposomal and standard NMN formulations, you can see that they have different pros and cons that affect how different market groups decide what to buy. Knowing these differences helps you make smart decisions about product creation and how to position yourself in the market.
Absorption and Bioavailability Profiles
The majority of clinical studies on NMN have used standard powder or capsule forms, and they have repeatedly shown that NAD+ levels rise even at low doses of around 250 mg. Based on these results, it seems that natural absorption pathways are already making conventional NMN bioavailable in a real way. Because NMN dissolves in water and has its own transporter systems, it has natural absorption benefits that make liposomal increase less necessary.
Liposomal NMN supporters say that encapsulation technology makes the drug more bioavailable by protecting it during transport and improving how cells take it in. But there haven't been many direct studies that compare liposomal and normal NMN, so it's still not clear what the real-world benefits of absorption are.
Formulation Flexibility and Applications
Standard NMN can be easily added to capsules, tablets, powders, and other healthy food products, giving formulators a lot of options. This substance dissolves easily in water, which makes production easier and ensures accurate dosing across all product formats.
The encapsulated version has special benefits in some situations, especially when it comes to transdermal cosmetics, where lipid carriers help the ingredients get deeper into the skin. The taste-masking qualities also make it possible to make liquid supplements and functional drinks that taste better and appeal to a wider range of consumers.
Cost-Benefit Analysis
The cost of each delivery method must be weighed against any possible market benefits by procurement managers. Standard NMN usually has lower prices for raw materials and easier manufacturing needs, which helps with competitive pricing strategies in mass-market uses.
Liposomal formulations cost more, but the higher prices may be worth it if the formulations improve the way the product is positioned, the user experience, or the effectiveness. A lot of the time, the choice is based on the brand's positioning strategies, its target market segments, and its competitors.
How to Choose the Right NMN Supplement for Your Business Needs?
To choose the right NMN formulations, you have to carefully look at a lot of different factors, such as the needs of your target market, your manufacturing skills, regulatory issues, and your goals for competitive positioning. When making a decision, scientific data must be weighed against long-term business goals and the ability to make money.
Quality Assessment and Supplier Evaluation
A strict evaluation of a supplier starts with checking that they have the right manufacturing certifications, such as ISO, GMP, and HACCP compliance. In order for B2B partnerships to work, these standards make sure that product quality is always the same, that regulations are followed, and that the supply chain is reliable. Facilities should be inspected, batch testing methods should be followed, and documentation should be looked over to make sure the supplier can do what they say they can do.
Analytical testing skills for liposomal NMN supplement are important criteria for evaluation, especially for complicated formulations like liposomal NMN. To make sure that products meet performance and shelf-life standards, suppliers must show that they are good at particle size analysis, encapsulation efficiency tests, and stability validation.
Application-Specific Considerations
For different uses in the market, different formulation methods are needed that meet specific performance needs and meet customer expectations. Nutraceutical businesses that are making high-end products for long life may benefit from liposomal technology's better positioning and possible bioavailability benefits.
Manufacturers of functional foods and drinks often put solubility, stability, and taste characteristics ahead of more advanced delivery methods. In these situations, standard NMN might give the best results while still being cost-effective and easy to make.
Regulatory and Compliance Factors
To get around in the regulatory landscape, you need to carefully think about safety statistics, claims support, and market-specific requirements. Standard NMN has been the subject of a lot of clinical research and has safety profiles that have been created. These profiles help the regulatory approval process in many places.
Liposomal products might need more safety information and claims to be backed up, especially when it comes to claims of better absorption. Procurement teams need to make sure that sellers give enough regulatory support and paperwork to make entering the market easier and keeping up with compliance easier.
Real-World Application and Procurement Strategies
To successfully buy liposomal NMN, you need to plan ahead and think about things like your relationships with suppliers, how to handle your inventory, and how to position your business in the market. Understanding the problems and chances of implementation in the real world helps people make better choices about purchases and build stronger partnerships.
Market Positioning and Differentiation
Companies that use liposomal technology often market their goods as high-end options with better bioavailability and a better user experience. This positioning approach helps businesses make more money by appealing to health-conscious customers who are willing to spend more on more advanced formulations.
Case studies from the supplement industry for liposomal NMN supplement show how liposomal goods can be strategically positioned as next-generation solutions to help them break into new markets. These examples show how important it is to have full marketing support and educational material that tells target audiences how technology can help them.
Supply Chain Optimization
Diversifying suppliers and managing relationships with them are important parts of good buying strategies that make sure there are always supplies and quality standards. Long-term relationships with reputable manufacturers give you access to technical support, help with developing new products, and better pricing structures that put you ahead of the competition.
When managing inventory, things to think about are the need for steadiness, the best way to extend the shelf life of items, and how accurate it is to predict demand. Liposomal formulations might need special ways to be stored and handled, which can raise the cost of transportation and make operations more difficult.
Technical Support and Innovation
Leading suppliers offer a wide range of professional support, such as help with formulating, testing for stability, and regulatory advice. These services are very helpful for businesses that want to come up with new products and figure out how to meet the needs of a complicated market.
Working together gives you access to new technologies and better formulations that give you a competitive edge in markets that are changing quickly. Regularly talking to providers keeps you up to date on changes in the industry and chances to save money.
Conclusion
Which one to use - liposomal NMN or standard NMN - really relies on your business goals, the needs of your target market, and how you want to position yourself in the market. Even though liposomal technology might be better in terms of bioavailability and formulation flexibility, normal NMN has been shown to work, is safe, and is cheaper. When making procurement choices, marketing claims should not be more important than scientific evidence, following the rules, and the possibility of a long-term partnership. For implementation to go smoothly, suppliers must be carefully evaluated, quality must be thoroughly evaluated, and the overall business goals must be strategically aligned with the implementation goals to ensure the best market success and customer satisfaction.
FAQ
Is liposomal NMN safer than standard NMN for commercial applications?
When made according to quality standards, both liposomal and standard NMN formulations show similar safety profiles. A lot of clinical study has been done on standard NMN to show that it is safe at different dose levels. Liposomal formulations use phospholipid carriers that are generally thought to be safe (GRAS) and don't pose many other safety issues. No matter what delivery method is used, procurement managers should give more weight to suppliers who have complete safety paperwork and the right regulatory certifications.
What dosage guidelines apply to commercial liposomal NMN products?
The recommended daily dose for both normal and liposomal formulations is usually between 250 mg and 500 mg. Standard NMN at these dose levels has been used in most of the clinical studies that show it works. It's possible that liposomal formulations need lower doses because they are more bioavailable, but specific dosing standards should be set by doing bioavailability studies on each product and talking to regulators.
Can liposomal NMN be customized for private labeling applications?
Most trustworthy companies that make liposomal NMN give you a lot of ways to customize it, such as private labeling, unique formulations, and different ways to package it. Customization options usually include finding the best particle size, changing the quantity, and mixing ingredients that work well together. Different suppliers have different minimum order quantities and wait times, so it's important to plan ahead for when you need to make custom products.
Partner with Bolin Biotechnology for Premium Liposomal NMN Solutions
Bolin Biotechnology stands as your trusted liposomal NMN manufacturer, combining twelve years of plant extract expertise with cutting-edge delivery technologies. Our state-of-the-art facilities maintain ISO, GMP, and HACCP certifications, ensuring consistent quality and regulatory compliance for your product development needs. We specialize in customized formulations, flexible MOQ arrangements, and comprehensive technical support that accelerates your market entry timeline. Our experienced team provides end-to-end solutions from initial concept through commercial production, backed by rigorous quality control and extensive regulatory documentation. Contact our specialist team at sales1@bovlin.com to explore exclusive B2B pricing, sample requests, and partnership opportunities that elevate your product portfolio with scientifically-backed NMN solutions.

References
Mills, K.F., Yoshida, S., Stein, L.R., Grozio, A., Kubota, S., Sasaki, Y., Redpath, P., Migaud, M.E., Apte, R.S., Uchida, K. and Yoshino, J. (2016). Long-term administration of nicotinamide mononucleotide mitigates age-associated physiological decline in mice. Cell metabolism, 24(6), pp.795-806.
Yoshino, J., Baur, J.A. and Imai, S.I. (2018). NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell metabolism, 27(3), pp.513-528.
Shade, C. (2016). Liposomes as advanced delivery systems for nutraceuticals. Integrative Medicine: A Clinician's Journal, 15(1), p.33.
Akbarzadeh, A., Rezaei-Sadabady, R., Davaran, S., Joo, S.W., Zarghami, N., Hanifehpour, Y., Samiei, M., Kouhi, M. and Nejati-Koshki, K. (2013). Liposome: classification, preparation, and applications. Nanoscale research letters, 8(1), pp.1-9.
Poddar, N.K., Maurya, A., Saxena, V., Ansari, V.A., Chaudhary, A., Kant, R. and Das, M. (2021). Enhanced bioavailability and tissue distribution of resveratrol by encapsulation in liposomes. International journal of pharmaceutics, 609, p.121146.
Grozio, A., Mills, K.F., Yoshino, J., Bruzzone, S., Sociali, G., Tokizane, K., Lei, H.C., Cunningham, R., Sasaki, Y., Migaud, M.E. and Imai, S.I. (2019). Slc12a8 is a nicotinamide mononucleotide transporter. Nature metabolism, 1(1), pp.47-57.











