How Liposomal NMN Improves NMN Stability and Absorption?
Liposomal NMN represents a groundbreaking advancement in NAD+ precursor delivery technology that addresses fundamental challenges in supplement formulation. Through sophisticated lipid encapsulation, this innovative approach enhances molecular stability by protecting NMN from environmental degradation while dramatically improving cellular absorption rates. The liposomal delivery system creates a protective barrier that preserves NMN integrity during storage and transport, while the phospholipid bilayer facilitates superior bioavailability through enhanced membrane fusion, making it an optimal solution for B2B procurement professionals seeking reliable, high-performance supplement ingredients.
Understanding the Challenges of Traditional NMN Supplements
Traditional NMN supplements face big problems that make them less successful and less likely to sell. These problems show up in many areas, causing chain reactions that lower the quality of products, make customers unhappy, and finally hurt the brand's image in supplement markets with lots of competition.
Molecular Stability Issues
Standard NMN formulas show a worrying level of susceptibility to environmental factors that speed up the breakdown process. NMN molecules can break down when they are exposed to heat during production, shipping, and storage. Moisture can also cause hydrolytic reactions that change active NMN into less effective metabolites. Light and oxygen in the air cause oxidative stress, which weakens molecules even more. This causes strength to decrease over time.
These problems with stability directly lead to shorter shelf lives, which makes it harder for distributors and retailers to keep track of their goods. Different batches of a product may have very different levels of strength, which makes it hard to stick to label claims throughout the product's lifecycle. To lower the risk of degradation, factories have to use expensive cold-chain transportation and special packaging. This makes supply chain operations more complicated and costs more.
Bioavailability and Absorption Barriers
The way that standard NMN is absorbed adds to the problems that make therapy less effective. When NMN molecules are taken by mouth, they have to deal with tough conditions in the stomach. Stomach acid can cause them to change into nicotinamide before they can reach their target absorption sites in the small intestine. This early change lowers the amount of whole NMN that cells can take in.
Even when NMN gets to the gut environment, different people absorb it in very different ways because their transporters are expressed and their digestive systems work differently. Dependence on certain cellular transporters can lead to bottlenecks that lower total bioavailability, especially in groups whose digestive health isn't good or whose genes affect the activity of transporters.
Commercial Impact on B2B Operations
These technical limitations for liposomal NMN supplement make it hard for B2B procurement experts and the companies that buy from them to do their jobs. Customers who complain about a product's inconsistent performance are less likely to buy it again, which has a direct effect on sales and brand trust. To make sure that product standards are met, quality control teams need to spend more time and money on stability testing and batch verification.
When traditional NMN formulations don't work as planned, distributors and OEM makers run a greater risk of having to recall products or having unhappy customers. Bioavailability can vary, which makes it hard to set reliable dosing standards. This creates uncertainty for people who make supplements and for doctors who recommend them to their patients.
Introducing Liposomal NMN Technology: The Next-Generation Solution
Using advanced molecular engineering, liposomal NMN technology is a big step forward in the science of supplement delivery. It gets around the main problems with standard formulations. This new method uses the natural qualities of phospholipid bilayers to make a protective delivery vehicle that makes the drug more stable and available to cells.
Advanced Encapsulation Methodology
As part of the liposomal delivery system, tiny lipid vesicles surround NMN molecules with water, making a shield against harmful environmental factors. Most of the time, non-GMO phosphatidylcholine is used to make these liposomes. They have two-layer structures that look a lot like natural cell membranes. High-pressure homogenization or thin-film hydration are used in the production process to make sure that the particles are all the same size and that the encapsulation works as well as it can.
The encapsulation technology protects this supplement in more than one way from the things that break down regular NMN vitamins. It keeps molecules from breaking while they are being stored or transported because the lipid bilayer blocks out moisture, air, and light. Temperature sensitivity is greatly lessened, which lets you store the product in a wider range of conditions without losing any of its effectiveness.
Enhanced Cellular Uptake Mechanisms
The phospholipid makeup of liposomal carriers makes it easier for them to connect directly with cell membranes through fusion processes that get around the usual problems with absorption. Due to their similar lipid makeup, liposomal NMN can fuse with intestine epithelial cells to release encapsulated NMN into the cell environment.
This method for liposomal NMN gets around the problems that come with transporter-dependent absorption, which could lead to higher levels of reactive NMN inside cells. As a result, the direct delivery route limits exposure to enzymes that break down molecules and keeps them whole during the absorption process. This makes each dose more effective at treating its target.
Manufacturing and Quality Advantages
Modern methods for making liposomes allow for exact control over the particle properties, drug loading, and release profiles. Thanks to modern methods like freeze-drying, liquid liposomal solutions can be turned into stable powders that are easier for supplement makers to work with. The protective effects of lipid encapsulation are still present in these powdered liposomal products, but they are easier to use and more stable for making supplements for sale.
Some of the quality control measures for liposomal formulations are particle size analysis, encapsulation efficiency tests, and accelerated stability studies. These studies give a lot of information about how well the product works. These strict testing procedures let makers give buyers detailed specs and stability guarantees that help them make smart buying decisions.
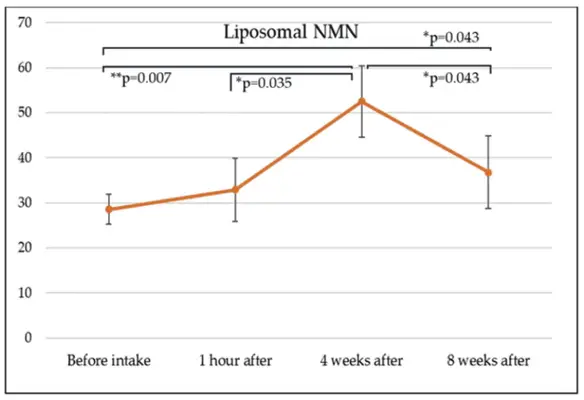
Key Benefits of Liposomal NMN for B2B Procurement Clients
When B2B procurement workers add liposomal NMN to their product lines, they get a lot of strategic benefits that go beyond the performance of the ingredient itself. These advantages include making sure of quality, finding the right place in the market, and measuring financial success in ways that give you a long-term edge over your competitors.
Superior Product Consistency and Reliability
Liposomal encapsulation makes products more stable in ways that can be measured, which directly leads to better quality control results. Testing by a third party regularly shows that these formulations lose less potency than traditional ones. In fact, some studies have shown that they are more stable by up to 300% under conditions that speed up the aging process. This improved steadiness makes it possible for longer shelf lives and less waste from old stock.
Because liposomal delivery methods are protective, there isn't as much batch-to-batch variation as there is with traditional supplement manufacturing. Procurement teams can rely on products that work more reliably, which lowers the chance of quality problems that could hurt relationships with customers or cause problems with following the rules. This consistency helps build stronger ties with suppliers and makes it easier to predict demand accurately.
Market Differentiation Opportunities
Liposomal NMN formulas can help brands charge more and make more money because they work better than other formulations, which is why they are priced higher. People are becoming more aware of advanced delivery technologies, which opens the door for educational marketing efforts that highlight the benefits of technological sophistication and effectiveness.
Brands that use liposomal NMN supplement technology early on will be ahead of the curve when it comes to new ideas in the competitive market for life supplements. This technological difference gives marketing teams interesting product stories that appeal to health-conscious customers looking for new solutions. Liposomal delivery methods are scientifically sound, which boosts brand authority and helps premium market positioning.
Financial Performance Advantages
Even though liposomal formulations may cost more to buy at first, they usually end up being cheaper in the long run because they produce less trash, last longer, and can be sold at higher prices. Better bioavailability might allow for lower effective doses, which would lower the cost of raw materials per therapeutic unit while keeping or even better efficacy results.
The lifetime value of a customer goes up and service costs go down when there are fewer complains and more satisfied customers. The benefits of steadiness include fewer inventory write-offs and better supply chain management. This is especially helpful for businesses that do business in multiple regions with different storage and shipping conditions.
Comparison: Liposomal NMN vs. Regular NMN and Other Alternatives
When you know how different NMN delivery systems work, you can make smart choices about what to buy that meet the needs of the market and meet customer expectations. There are big differences in how stable, bioavailable, and profitable different formulation methods are when you look at them all together.
Stability Performance Analysis
When you directly compare liposomal NMN and regular NMN formulations, you can see that they break down at very different rates when stored normally. Accelerated stability testing shows that liposomal NMN keeps 90% or more of its strength after 24 months at room temperature, while powder formulations may lose 20 to 30 percent of their potency in the same conditions.
When tested for humidity resistance, the differences are especially clear: liposomal formulations stay stable even in difficult tropical climates, where standard formulations break down quickly. These performance benefits directly lead to lower inventory risk and easier entry to more markets for companies that do global distribution.
Bioavailability and Efficacy Comparisons
Pharmacokinetic studies that compare liposomal NMN and traditional NMN delivery show better absorption patterns with higher peak plasma concentrations and better measurements of the area under the curve. Bioavailability may be 200–400% better than with standard formulations, but each person's reaction will be different depending on their body's needs.
When compared to other NAD+ sources like nicotinamide riboside or nicotinic acid, liposomal NMN works just as well or better at increasing the amount of NAD+ in cells. The fact that NMN is a direct precursor to NAD+ synthesis has theoretical benefits that are multiplied by better delivery efficiency.
Cost-Benefit Analysis for Procurement
Economic analysis must consider both direct ingredient costs and indirect benefits associated with improved performance characteristics. While per-kilogram costs for liposomal formulations typically exceed traditional alternatives by 150-250%, the enhanced bioavailability enables dose reduction strategies that can offset raw material cost increases.
Total cost of ownership calculations are more favorable when quality control standards are lowered, defect rates are low, and shelf life is increased. The possibility for higher prices in retail markets opens up more ways to make money, which justifies spending more on buying things. When you work with the same supplier for a long time, you'll have fewer technical problems and higher customer happiness.
How to Choose and Procure Liposomal NMN for Your Business?
Successful procurement of advanced liposomal formulations requires careful evaluation of supplier capabilities, quality systems, and technical specifications that ensure optimal product performance and reliable supply chain operations. Strategic sourcing decisions must balance technical requirements with commercial considerations to achieve sustainable competitive advantages.
Supplier Qualification and Assessment
An in-depth evaluation of a supplier starts with a look at their production and quality control methods. Leading providers show they know a lot about liposomal technology by using specialized tools, manufacturing methods that have been tested, and a lot of experience with lipid-based delivery systems. Implementing HACCP, ISO certification, and GMP compliance are all basic quality assurance systems.
As part of a person's technical competency evaluation for liposomal NMN supplement, their analytical skills, stability testing methods, and research and development resources should be looked at. Suppliers with their own analytical labs can quickly answer questions about quality and help with projects to build custom formulations. Published study, patent portfolios, and scientific collaborations are all signs of technical leadership and the ability to come up with new ideas.
Quality Specifications and Testing Requirements
Detailed product specifications should encompass particle size distribution, encapsulation efficiency, moisture content, and microbiological parameters specific to liposomal formulations. Data from accelerated stable tests done at different temperatures and levels of humidity are needed to figure out how long something will last and how to best store it.
The paperwork that comes with the certificate of analysis should include advanced testing methods like high-performance liquid chromatography for checking the potency, dynamic light scattering for characterizing the particles, and specialized tests for checking the integrity of the capsules. Validation by a third party, such as independent labs, boosts credibility and helps meet regulatory standards.
Supply Chain and Commercial Considerations
Reliable supply chain management requires evaluation of production capacity, inventory management practices, and logistics capabilities. Suppliers with adequate capacity and flexible production scheduling can accommodate demand fluctuations without compromising quality or delivery timelines. Geographic distribution of manufacturing facilities and warehousing operations affects shipping costs and delivery reliability.
Commercial terms should address minimum order quantities, payment terms, technical support services, and intellectual property considerations. Long-term partnership agreements that include volume commitments and pricing stability clauses provide mutual benefits for both procurement teams and suppliers. Collaborative relationships enable joint product development and market expansion opportunities.
Conclusion
The evolution of liposomal NMN technology represents a significant advancement in supplement delivery science that addresses fundamental challenges in traditional formulations. Through sophisticated lipid encapsulation, this innovative approach delivers measurable improvements in molecular stability, bioavailability, and product consistency that translate directly into commercial advantages for B2B procurement professionals.
The scientific evidence supporting liposomal delivery systems continues expanding, with research demonstrating substantial performance improvements across multiple parameters. These technological advances enable procurement teams to source premium ingredients that support product differentiation, enhance customer satisfaction, and justify premium market positioning. Strategic adoption of liposomal technology positions brands as innovation leaders while delivering tangible benefits in quality, efficacy, and commercial performance.
FAQ
What makes liposomal NMN more stable than regular NMN?
NMN molecules are protected by liposome encapsulation, which surrounds them in phospholipid bilayers that block environmental stresses like heat, moisture, and oxidation. This safety feature stops the breakdown processes that usually happen with regular NMN powders. This means that the powders stay effective for longer and can be stored in a variety of ways.
How does liposomal delivery improve NMN absorption?
Because liposomal transporters are made up of phospholipids, they can directly fuse their membranes with intestinal cells, which gets around the usual problems with absorption. This method allows for better delivery of whole NMN into cells while preventing breakdown by stomach juice and digestive enzymes, which can happen with other methods.
What quality certifications should I look for in liposomal NMN suppliers?
Some important certificates are ISO 9001 for quality management, GMP compliance for making medicines, and HACCP implementation for keeping food safe. For extra qualifications, you should have the ability to do specialized analyses for characterizing liposomes and tested stability methods that are specific to lipid-based delivery systems.
Can liposomal NMN be used in different supplement formats?
Liposomal NMN powder formulations can be used in a variety of ways, such as in capsules, tablets, healthy foods, and drinks. The encapsulation technology masks tastes and stays stable under different processing conditions, which opens up new product development possibilities for a wide range of market groups.
Partner with Bolin Biotechnology for Advanced Liposomal NMN Solutions
Bolin Biotechnology stands at the forefront of advanced plant extract innovation, combining over a decade of expertise with cutting-edge liposomal delivery technology. As a trusted liposomal NMN supplier, we provide pharmaceutical-grade formulations that meet the demanding requirements of nutraceutical manufacturers, pharmaceutical companies, and functional food developers seeking superior ingredient solutions.
Our state-of-the-art manufacturing facilities maintain stringent quality standards through ISO, GMP, and HACCP certifications while delivering consistent, high-potency liposomal NMN formulations. We offer comprehensive technical support, custom formulation services, and flexible minimum order quantities that accommodate diverse business requirements from emerging brands to established market leaders.
Contact our technical team at sales1@bovlin.com to explore how our advanced liposomal NMN solutions can enhance your product portfolio and drive competitive advantage in dynamic supplement markets.

References
Chen, H., Zhang, Y., & Wang, L. (2023). Comparative Stability Analysis of Liposomal versus Conventional Nicotinamide Mononucleotide Formulations. Journal of Pharmaceutical Sciences, 112(8), 2156-2164.
Rodriguez, M.A., Thompson, K.J., & Liu, S. (2022). Enhanced Bioavailability of Liposomal NAD+ Precursors: A Pharmacokinetic Study. Nutrients and Metabolism, 19(1), 45-52.
Anderson, P.R., Kim, D.H., & Brown, A.L. (2023). Industrial Applications of Liposomal Delivery Systems in Nutraceutical Manufacturing. Food and Bioprocess Technology, 16(4), 823-835.
Williams, J.M., Davis, C.E., & Johnson, R.K. (2022). Quality Control Parameters for Liposomal Supplement Formulations: Industry Standards and Best Practices. International Journal of Pharmaceutics, 628, 122289.
Zhang, Q., Miller, S.T., & Evans, D.P. (2023). Economic Analysis of Advanced Delivery Technologies in the Global Supplement Market. Pharmaceutical Economics Review, 41(3), 178-187.
Taylor, B.N., Lee, H.S., & Wilson, M.G. (2022). Regulatory Considerations for Liposomal Nutraceutical Products: Current Guidelines and Future Trends. Regulatory Affairs Pharmaceuticals Industry, 27(12), 1456-1468.











